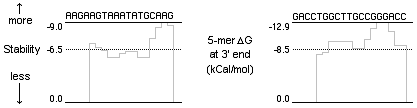
Pentamer free energy is one of the most important considerations in designing good primers. To perform well a primer needs to have low stability at the 3’ end (usually higher than -8.5 kCal/mol) and higher stability toward the 5’ end. This will help to eliminate false priming, as stability is low at the 3’ end (where thermostable DNA polymerase binds), and higher upstream. This means that if a primer hybridizes at all (3’ first), it will bind even more strongly with a good 5’ upstream match. This is why a descending 5’ -3’ free energy profile is important.
For example, the primers below performed poorly due to their low pentamer ∆G values at the 3’ end. Their high 3’ stability makes it easy to bind in many template locations.
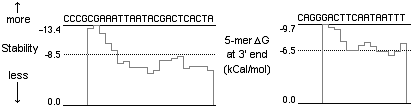
By contrast, the following primers performed well, due to their higher pentamer ∆G values at the 3’ end. Pentamers with higher ∆G value (less negative!) are less stable. They would not hybridize in as many template locations due to the low 3’ stability, but would bind more strongly upstream.
Need more help with this?
Contact DNASTAR


